
 Credit: X-ray spectra/NASA/CXC/MPI/M.Brusa et al.
Credit: X-ray spectra/NASA/CXC/MPI/M.Brusa et al.
Iron Dating
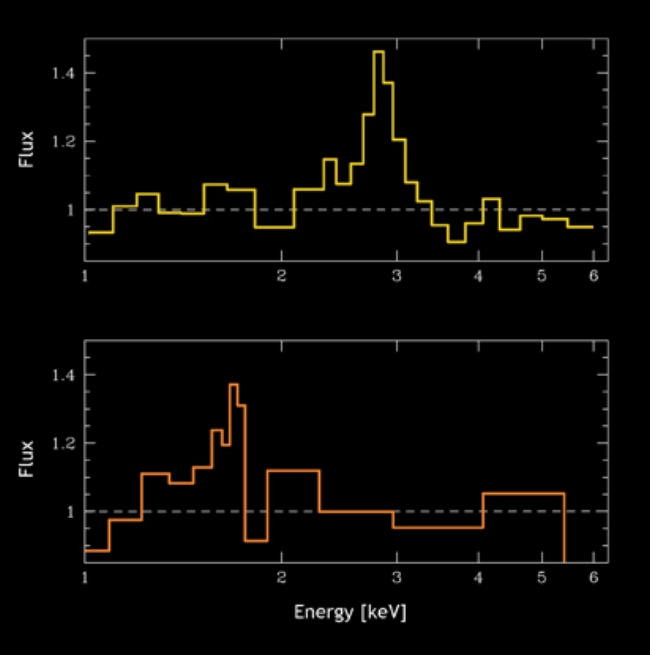
How does the chemistry of the Universe evolve? The early Universe consisted of hydrogen and helium and very little else. The present Universe has substantial amounts of complex atoms like carbon, nitrogen, oxygen and even heavier elements. Astronomers know that most of these elements are cooked up in stars, or by cosmic explosions, but exactly when most of the elements we see today were created is a mystery. But it's a mystery astronomers are trying to solve. Using observations of black holes from the Chandra X-ray Observatory "Deep Field", astronomers have begun to probe this issue. The image above shows spectra from 2 sets of black holes, one set (upper panel) at a distance of about 9 billion light years, and another set (lower panel) from about 11 billion light years distant. The "bump" of emission in both panels is produced by iron atoms near the black holes, and the strength of these "bumps" is proportional to the abundance of iron. This lets astronomers constrain the amount of iron in the Universe at 9 and 11 billion years ago. Somewhat surprisingly, the amount of iron at these two times is fairly similar. This might mean that most of the iron in the Universe was produced earlier than 11 billion years ago, with relatively little creation of iron since.
Last Week *
HEA Dictionary * Archive
* Search HEAPOW
* Education
Each week the HEASARC
brings you new, exciting and beautiful images from X-ray and Gamma ray
astronomy. Check back each week and be sure to check out the HEAPOW archive!
Page Author: Dr. Michael F. Corcoran
Last modified Monday, 26-Feb-2024 17:35:17 EST


